Abstract
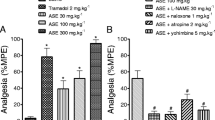
The antinociceptive action of quercetin, a common bioactive flavonoid present in many medicinal plants, was assessed in different models of chemical and thermal nociception in mice. Quercetin (10–60 mg/kg, i.p. or 100–500 mg/kg, p.o.) dose-dependently inhibited nociceptive behavior in the acetic acid-induced pain test. Moreover, quercetin (10–60 mg/kg, i.p.) inhibited both phases of formalin-induced pain, with ID50 values of 374.1 (68.0–402.0) mmol/kg and 103.0 (45.0–201.0) mmol/kg, for the neurogenic and inflammatory phases, respectively. Quercetin (10–60 mg/kg) also inhibited the nociception induced by glutamate and capsaicin by 68.2% and 75.5%, respectively. Its analgesic action was significantly reversed by p-chlorophenylalanine methyl ester, katanserin, methysergide, a GABAA antagonist (bicuculline), or a GABAB antagonists (baclofen). Its action was also modulated by tachykinins, but was not affected by adrenal-gland hormones. Furthermore, the antinociceptive effects did not result from muscle-relaxant or sedative action. Together, these results indicate that quercetin produces dose-related anti-nociception in several models of chemical pain, through mechanisms that involve interaction with L-arginine-nitric oxide, serotonin, and GABAergic systems. These results confirm and extend other investigations on the analgesic effect of quercetin and its mechanisms of action.
Similar content being viewed by others
References
Anjaneyulu, M. and Chopra, K., Quercetin attenuates thermal hyperalgesia and cold allodynia in STZ-induced diabetic rats. Indian J. Exp. Biol., 42, 766–769 (2004).
Beirith, A., Santos, A. R. S., and Calixto, J. B., Mechanism underlying the nociception and paw edema caused by injection of glutamate into the mouse paw. Brain Res., 924, 219–228 (2002).
Carter, G. T. and Sullivan, M. D., Antidepressants in pain management. Curr. Opin. Investig. Drug, 3, 454–458 (2002).
Cechinel Filho, V., Santos, A. R., De Campos, R. O., Miguel, O. G., Yunes. R. A., Ferrari, F., Messana, I., and Calixto, J. B., Chemical and pharmacological studies of Phyllanthus caroliniensis in mice. J. Pharm. Pharmacol., 48, 1231–1236 (1996).
Choi, S. S., Lee, J. K., and Suh, H. W., Antinociceptive profiles of aspirin and acetaminophen in formalin, substance P and glutamate pain models. Brain Res., 921, 233–239 (2001).
Collier, H. D. J., Dinnin, L. C., Johnson, C. A., and Schneider, C., The abdominal response and its suppression by analgesic drugs in the mouse. Br. J. Pharmacol., 32, 295–310 (1968).
Comalada, M., Camuesco, D., Sierram S., Ballesterm I., Xausm J., Galvez. J., and Zarzuelo, A., In vivo quercitrin anti-inflammatory effect involves release of quercetin, which inhibits inflammation through down-regulation of the NF-kappa B pathway. Eur. J. Immunol., 35, 584–592 (2005).
Eddy, N. B. and Leimbarck, D., Synthetic analgesics II. Dithienylbutenyl and dithienylbutylamines. J. Pharm. Exp. Ther., 107, 385–393 (1953).
Franek, M., Vaculín, S., and Rokyta, R., GABAB receptor agonist baclofen has non-specific antinociceptive effect in the model of peripheral neuropathy in the rat. Physiol. Res., 53, 351–355 (2004).
Hizue, M. A., Pang, C.A., and Yokoyama, M. B., Involvement of N-methyl-D-aspartate-type glutamate receptor [varepsilon]1 and [varepsilon]4 subunits in tonic inflammatory pain and neuropathic pain. Neuroreport., 16, 1667–1670 (2005).
Hunskaar, S., Fasmar, O. B., and Hole, K., Formalin test in mice: a useful technique for evaluating mild analgesics. J. Neurosci. Methods, 14, 69–76 (1985).
Hunskaar, S. and Hole, K., The formalin test in mice: dissociation between inflammatory and non-inflammatory pain. Pain, 30, 103–114 (1987).
Kauer, R., Singh, D, and Chopra, K., Participation of alpha2 receptors in the antinociceptive activity of quercetin. J. Med. Food, 4, 529–532 (2005).
Kris-Etherton, P. M., Lefevre, M., Beecher, G. R., Gross, M. D., Keen, C. L., and Etherton, T. D., Bioactive compounds in nutrition and health-research methodologies for establishing biological function: the antioxidant and anti-inflammatory effects of flavonoids on atherosclerosis. Annu Rev. Nutr., 24, 511–538 (2004).
Kumazawa, T., Mizumura, K., Koda, H., and Fukusako, H. EP receptor subtypes implicate in the PGE2-induced sensitization of polymodal receptors in response to bradykinin and heat. J.Neurophysil., 75, 2361–2368 (1996).
Lee, B. H., Jeong, S. M., Lee, J. H., Kim, J. H., Yoon, I. S., Lee, J. H., Choi, S. H., Lee, S. M., Chang, C. G., Kim, H. C., Han, Y., Paik, H. D., Kim, Y., and Nah, S. Y., Quercetin inhibits the 5-hydroxytriptamine type 3 receptor-mediated ion current by interacting with pre-transmembrane domain I. Mol. Cells., 20, 60–73 (2005).
Liu, J., Yu, H., and Ning, X., Effect of quercetin on chronic enhancement of spatial learning and memory of mice. Scj. China C Life Sci., 49, 583–590 (2006).
Mackey, S. and Feinberg, S., Pharmacologic therapies for complex pain syndrome. Curr. Pain Headache Rep., 11, 38–43 (2007).
Manthey, J. A., Biological properties of flavonoids pertaining to inflammation. Microcirculation, 7, S29–S34 (2000).
Mantovani, M., Kaster, M. P., Pertile, R., Calixto, J. B., Rodrigues, A. L., and Santos, A. R., Mechanisms involved in the antinociception caused by melatonin in mice. J. Pineal Res., 41, 382–389 (2006).
Millan, M. J., Descending control of pain. Prog Neurobiol., 66, 355–474 (2002).
Moskaug, J. O., Carlsen, H., Myhrstad, M., and Blomhoff, R., Molecular imaging of the biological effects of quercetin and quercetin-rich foods. Mech. Ageing Dev., 125, 315–324 (2004).
Murota, K. and Terao, J., Antioxidative flavonoid quercetin: implication of its intestinal absorption and metabolism. Arch. Biochem. Biophys., 417, 12–17 (2003).
Naidu, P. S., Singh, A., and Kulkarni, S. K., D2-dopamine receptor and alpha2-adrenoreceptor-mediated analgesic response of quercetin. Indian J. Exp. Biol., 41, 1400–1404 (2003).
Neves, S. A., Freitas, A. L. P., Souza, B. W. S., Rocha M. L. A., Correia, M. V. O., Sampaio, D. A., and Viana, G. S. B., Antinociceptive properties in mice of a lectin isolated from the marine alga Amansia multifida Lamouroux. Braz. J. Med. Biol. Res., 40, 127–134 (2007).
Olszanecki, A., Gebska, A., Kozlovski, V. I., and Gryglewski, R. J., Flavonoids and nitric oxide synthase. J. Physiol. Pharmacol., 53, 571–584 (2002).
Okamoto, T., Safety of quercetin for clinical application. Int. J. Mol. Med., 16, 275–278 (2005).
Picq, M., Cheav, S. L., and Prigent, A. F., Effect of two flavonoid compounds on central nervous system: analgesic activity. Life Sci., 49, 1979–1988 (1991).
Riedel, W. and Neeck, G., Nociception, pain, and antinociception: current concepts. Z. Rheumatol., 60, 404–415 (2001).
Rodrigues, A. L, da Silva, G. L., Mateussi, A. S., Fernandes, E. S., Miguel, O. G., Yunes, R. A., Calixto, J. B., and Santos, A. R. S., Involvement of monoaminergic system in the antidepressant-like effect of the hydroalcoholic extract of Siphocampylus verticillatus. Life Sci., 70, 1347–1358 (2002).
Rylski, M., Duriasz-Rowinska, H., and Rewerski, W., The analgesic action of some flavonoids in the hot-plate test. Acta Physiol. Pol., 30, 385–388 (1979).
Sakurada, T., Katsumata, K., Yogo, H., Tan-No, K., Sakurada, S., and Kisara, K., The capsaicin test in mice for evaluating tachykinin antagonists in the spinal cord. Neuropharmacol., 31, 1279–1285 (1992).
Sakurada, T., Katsumata, K., Yogo, H., Tan-No, K., Sakurada, S., and Kisara, K., Antinociception induced by CP96,345, a non-peptide NK1-receptor antagonist, in the mouse formalin and capsaicin tests. Neurosci. Letter., 151, 142–145 (1993).
Santos, A. R. S., PhD Thesis, UFSC, Florianópolis, Brazil, 2001.
Santos, A. R. S. and Calixto, J. B., Further evidence for the involvement of tachycinin receptor subtypes in formalin test and capsaicin models of pain in mice. Neuropeptides, 31, 381–389 (1997).
Santos A. R. S., Vedana, E. M., and De Freitas, G. A., Antinociceptive effect of meloxicam, in neurogenic and inflammatory nociceptive models in mice. Inflamm. Res., 47, 302–307 (1998).
Santos, A. R., Gadotti, V. M., Oliveira, G. L., Tibola, D., Paszcuk, A. F., Neto, A., Spindola, H.M. De Souza, M. M., Rodrigues, A. L., and Calixto, J. B. Mechanisms involved in the anti-nociception caused by agmatine in mice. Neuropharmacol., 48, 1021–1034 (2005).
Schultke, E., Kendall, E., Kamencic, H., Ghong, Z., Griebel, R. W., and Juurlink B. H., Quercetin promotes functional recovery following acute spinal cord injury. J. Neurotrauma, 20, 583–591 (2003).
Theoharides, T. C., Treatment approaches for painful bladder syndrome/interstitial cystitis. Drugs, 6, 215–235 (2007).
Thomasset, S. C., Berry, D. P., Garcea, G., Marczylo, T., Steward, W. P., and Gescher, A. J., Dietary polyphenolic phytochemicals-promising cancer chemopreventive agents in humans? A review of their clinical properties. Int. J. Cancer, 120, 451–458 (2007).
Toker, G., Kupeli, E., Memisoglu, M., and Yesilada, E., Flavonoids with antinociceptive and anti-inflammatory activities from the leaves of Tilia argentea (silver linden). J. Ethnopharmacol., 95, 393–399 (2004).
Zimmermann, M., Ethical guidelines for investigations of experimental pain in conscious animals. Pain, 16, 109–110 (1983).
Yashpal, K., Fisher, K., Chabot, J. G., and Coderre, T. J., Differential effects of NMDA and group I mGluR antagonists on both nociception and spinal cord protein kinase C translocation in the formalin test and a model of neuropathic pain in rats. Pain, 94, 17–29 (2001).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Filho, A.W., Filho, V.C., Olinger, L. et al. Quercetin: Further investigation of its antinociceptive properties and mechanisms of action. Arch. Pharm. Res. 31, 713–721 (2008). https://doi.org/10.1007/s12272-001-1217-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-001-1217-2